International Regulatory
Product registrations with health authorities of the respective countries would be the key to advance in any international market. Over the years, we have gained an in-depth understanding of the regulatory framework of many countries to accelerate product registration dossiers.
At Advin, we have a dedicated & qualified team to handle various functions of regulatory affairs.
Our Regulatory Team can provide a complete support to register the Products in respective countries with consultation of client.
International Market
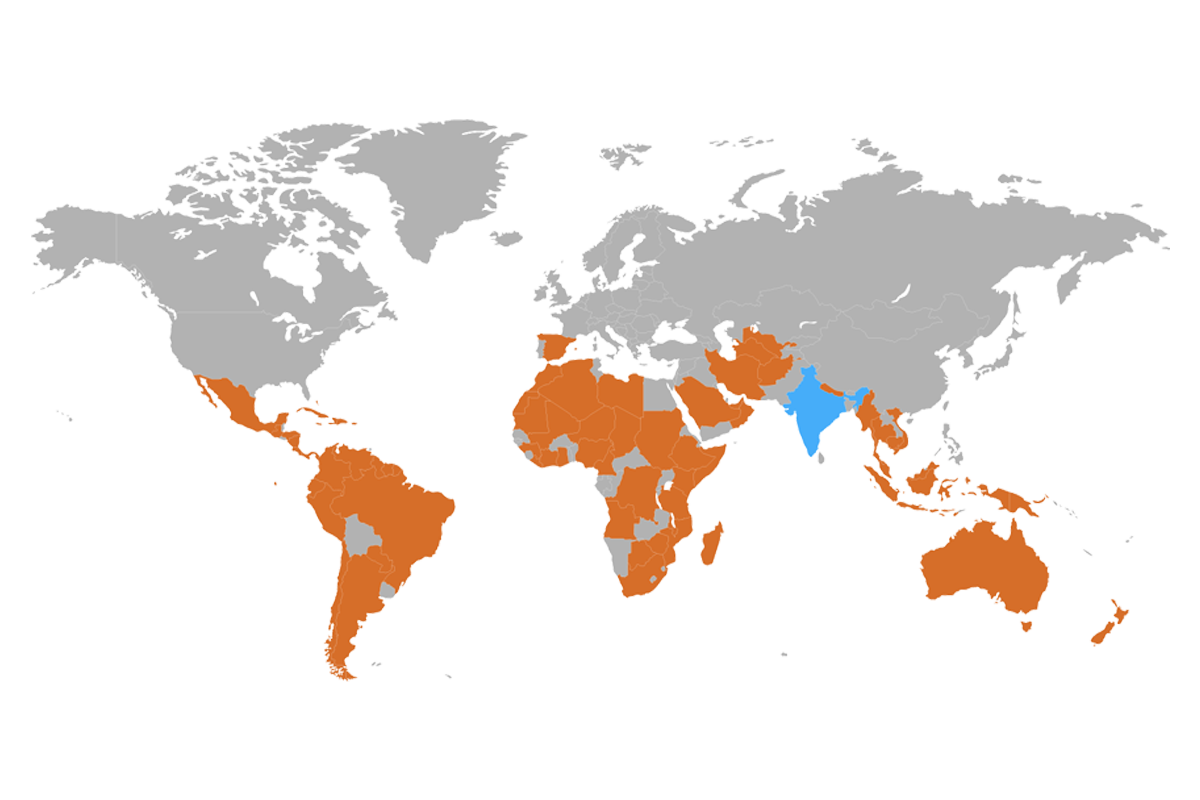
We export our products to Philippines, Guatemala, Honduras, Nepal, Pakistan, Sri Lanka, Azerbaijan, Indonesia, Malaysia, Dubai, Thailand, Maldives, Ghana, Yemen, Chad, Bolivia, Nicaragua.
